- Blog
- Large car model builder
- Custom faceless portraits
- Tire air compressor
- Toon boom studio vs animate
- Easy cat treats
- Macallan whiskey 12 double cask
- Endless sky map
- Repeating a unit cell in crystalmaker
- Hidden object game letters from nowhere 2
- Free student microsoft office 2016
- Free idvd themes
- Sally face game metal band epusode 1
- Format papier de infix pro
- Synology drive review
- Drakengard 3 zero icons
- Winmerge vs beyond compare
- Stellarium crashes on startup
- Scuttlebutt restaurant daytona beach
- Macos monterey release date 2021
- Star wars click and drag game
- Formz modx form validation reloads whole page
- Sumer civilization
- Json editor online chrome extension

In igneous rocks it occurs both as dispersed very small grains and as large crystals in pegmatites and it is also found as a detrital mineral in sediments. It occurs in metamorphic rocks such as amphibolites, eclogites and kimberlite xenoliths. Rutile (TiO 2) has a tetragonal structure (space group P4 2/ mnm.).
It has low reflectance in reflected light and the high hardness can cause difficulties during polishing that leave many surface scratches that are nevertheless an aid to identification. Corundum has a high hardness (9 on Mohs scale). There is often some zonation and alteration lamellae may be present. In thin section corundum has a high relief and it is generally colorless although tinges of the red of ruby or the blue of sapphire can sometimes be apparent. These unstable phases are β-Al 2O 3 (hexagonal), γ-Al 2O 3 (cubic), δ-Al 2O 3 (tetragonal or orthorhombic), κ-Al 2O 3 (orthorhombic or hexagonal), ε-Al 2O 3 (hexagonal), η-Al 2O 3 (cubic) and σ-Al 2O 3 (cubic). In addition to corundum (α-Al 2O 3) there are natural and synthetic Al 2O 3 phases that are not considered as polymorphs but are transitional stages between corundum and the Al-hydroxides. The effect of these impurities is to color the corundum, causing the red of ruby, the blue of sapphire and green, yellow, orange, pink, and brown varieties ( Fig. Most of these elements are usually only present in amounts up to a few hundred ppm but Cr can reach 2–3 wt% and Fe up to 1 wt%. Ĭorundum consists of nearly pure Al 2O 3 with trace to minor amounts of Cr, V, Fe, Ni, Ga, Ca and Mn whilst Na, Mg, Si, Co, Cd, Ga, S and Zr can also occur. Weldon, owned by J Ambalu, Amba Gem Corp, reproduced with permission from Hughes RW, Manorotkul W, and Hughes EB (2017) Ruby & Sapphire: A Gemologist's Guide.
#Repeating a unit cell in crystalmaker software
Oxford, England: CrystalMaker Software Ltd, (C) Reproduced with permission from Bowles JFW, Howie RA, Vaughan DJ, and Zussman J (2011) The Rock Forming Minerals, vol.
#Repeating a unit cell in crystalmaker for mac
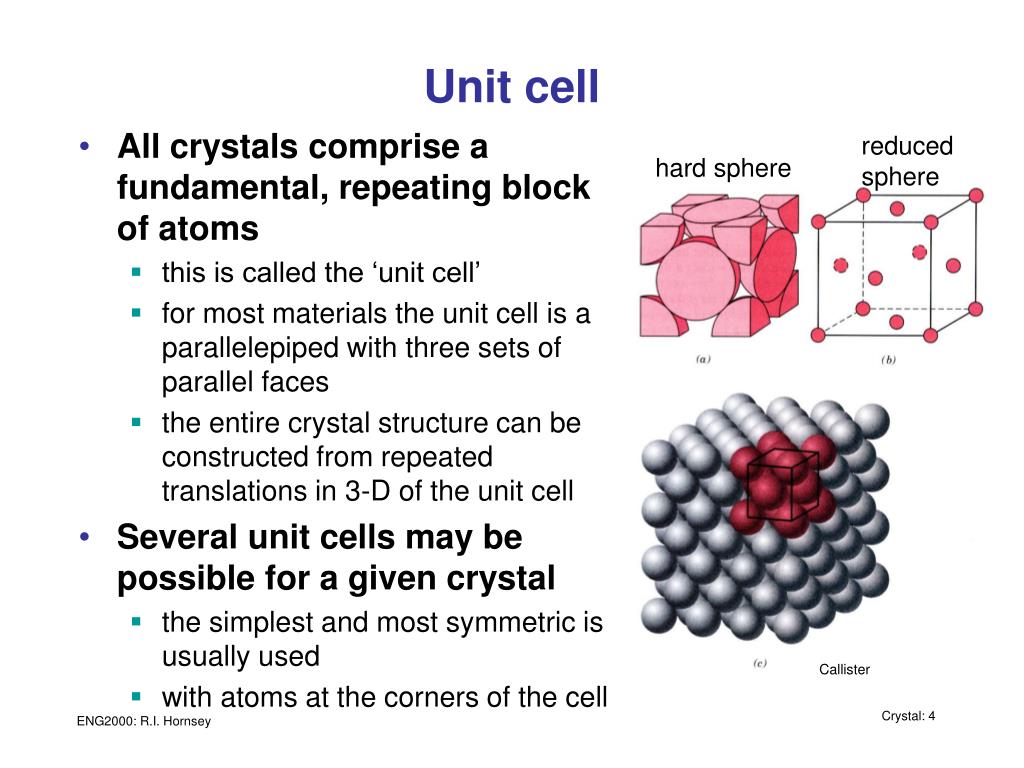
Crystal Structure Images Generated Using CrystalMaker®: A Crystal and Molecular Structures Program for Mac and Windows. (C) the color of corundum is determined by the proportions of the trace elements. (A) The unit cell of corundum viewed down the z axis, (B) the unit cell viewed down the x axis.
- Blog
- Large car model builder
- Custom faceless portraits
- Tire air compressor
- Toon boom studio vs animate
- Easy cat treats
- Macallan whiskey 12 double cask
- Endless sky map
- Repeating a unit cell in crystalmaker
- Hidden object game letters from nowhere 2
- Free student microsoft office 2016
- Free idvd themes
- Sally face game metal band epusode 1
- Format papier de infix pro
- Synology drive review
- Drakengard 3 zero icons
- Winmerge vs beyond compare
- Stellarium crashes on startup
- Scuttlebutt restaurant daytona beach
- Macos monterey release date 2021
- Star wars click and drag game
- Formz modx form validation reloads whole page
- Sumer civilization
- Json editor online chrome extension
